Four-membered carbocycles contain highly strained molecular structures and show unique conformation properties, which enable them to be important building blocks in organic synthesis and act as one of the fundamental units in a variety of biologically active substances.
Catalytic asymmetric synthesis of cyclobutanes and their derivatives developed rapidly, including [2+2] cycloadditions and skeletal rearrangements such as ring expansion or contraction. New approaches allowing access to enantioenriched four-membered ring scaffolds in a concise, highly selective, and diversified way are still challenging but highly desirable.
In a study published in Journal of the American Chemical Society, the research group led by Prof. FANG Xinqiang from Fujian Institute of Research on the Structure of Matter of the Chinese Academy of Sciences realized the first asymmetric transfer hydrogenation of cyclobutenediones in a highly regio- and stereoselective fashion.
The researchers found that the protocol delivers a series of synthetically and biologically important four-membered carbocycles including cyclobutenones, cyclobutanediols, cyclobutenediols, and cyclobutenols with up to 99% ee and >20:1 dr.
The highly functionalized products enable a series of further useful transformations and applications, including the asymmetric total synthesis of di-, tri-, and tetrasubstituted bioactive or pharmaceutically active molecules.
Systematic mechanistic studies revealed that the aryl rings in the substrates play a key role in achieving high regioselectivity. The rational selection of the two substituents of CBDs is crucial for obtaining high enantioselectivity.
Besides, the researchers executed the key hydrogenation transition state models, and the sequence of the double and triple hydrogenation processes by density functional theory(DFT) calculations.
This study provides a new choice for the diversified asymmetric synthesis of cyclobutane and its analogs and demonstrates the huge potential of ATH in achieving selective transformations of highly functionalized substrates.
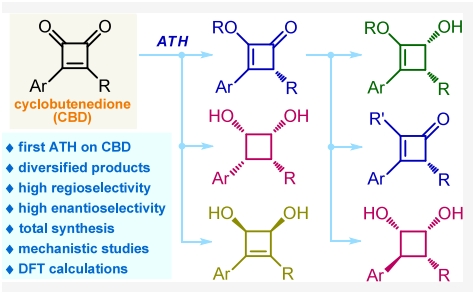
Asymmetric Transfer Hydrogenation of Cyclobutenediones (Image by Prof. FANG Xinqiang's group)
Contact:
Prof. FANG Xinqiang
Fujian Institute of Research on the Structure of Matter
Chinese Academy of Sciences
Email: xqfang@fjirsm.ac.cn