Harnessing solar energy through artificial photosynthesis is an attractive and sustainable approach to reducing global reliance on non-renewable fossil fuels and addressing environmental contamination. Solar-driven overall water splitting (OWS), particularly via photocatalysis, has become a dynamic field for converting solar energy into storable hydrogen (H₂) fuel. The primary challenge in achieving an efficient OWS process is designing photocatalysts that excel in light absorption, charge separation, and surface catalytic reactions.
Additionally, a significant issue is that the oxygen (O₂) produced during water splitting competes with hydrogen protons in reduction reactions. This reverse reaction consumes electrons and protons that are intended for hydrogen evolution reaction (HER), thereby diminishing the overall efficiency of the photocatalytic OWS process. Therefore, enhancing forward reactions while suppressing backward reactions is crucial for optimizing the photocatalytic OWS process.
In a study published in Angewandte Chemie Internation Edition, Prof. WANG Yaobing and his team from the Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, proposed using molecular engineering through methylation to inhibit the backward oxygen reduction reaction (ORR) in molecular photocatalysts, thereby achieving an efficient OWS process.
This approach was demonstrated using a representative sulfone-containing covalent organic framework (COF) with intrinsic photocatalytic hydrogen evolution reaction (HER) activity. Remarkably, the resulting methylated DS-COF exhibited significantly enhanced photocatalytic OWS performance, achieving a hydrogen production rate of 124.7 μmol h⁻¹ g⁻¹. This performance notably surpassed that of the methyl-free S-COF and ranks among the highest values reported for organic photocatalysts.
The researchers demonstrated that methylation enhances charge separation and extends the charge lifetime from 166.5 to 619.0 picoseconds, as evidenced by femtosecond transient absorption (fs-TA) spectra. This modification also contributes to the formation of electron-rich hydrogen adsorption sites (1.25 |e|) and electron-deficient oxygen adsorption sites (0.02 |e|). These effects are attributed to the increased polarization (from 9.69 to 10.90 Debye) between the triformylphloroglucinol donor and the sulfone acceptor, as revealed by density functional theory (DFT) calculations.
In-situ Fourier-transform infrared spectroscopy (FT-IR) and photocatalytic characterizations further showed that methylation promotes the forward hydrogen evolution reaction (HER) while inhibiting approximately 70% of the backward oxygen reduction reaction (ORR). From a molecular orbital perspective, this is explained by a significant peak shift in the antibonding direction (from -0.388 to -0.314) and an elevated energy barrier against O₂ adsorption and H₂O desorption, respectively. By simultaneously obstructing electron transfer and increasing the kinetic barrier for O₂ adsorption, the proton-coupled electron transfer (PCET) process of ORR is substantially suppressed.
This study presents a strategy for photocatalyst modification with high efficiency water splitting, which provides a molecular engineering perspective for the design of efficient OWS photocatalysts.
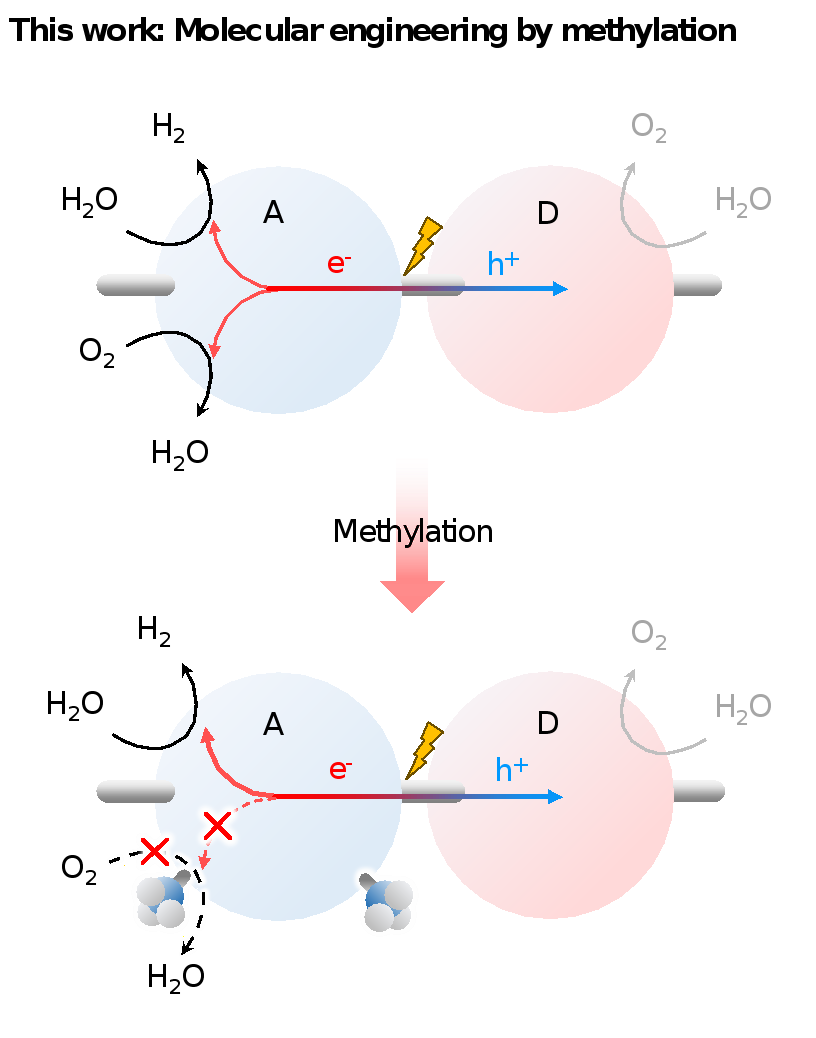
Schematic representation illustrating the modification strategy for enhanced photocatalytic OWS. (Image by Prof. WANG’s group)
Contact:
Prof. WANG Yaobing
Fujian Institute of Research on the Structure of Matter
Chinese Academy of Sciences
Email: wangyb@fjirsm.ac.cn