Large bone defects are notoriously difficult to repair because conventional scaffolds often ignite excessive inflammation and fail to build a functional vascular network. A successful repair requires a precise sequence: first calm inflammation, then grow blood vessels, and finally form new bone, yet most materials target only one step.
In a study published in Biomaterials, the researchers led by Prof. LIN Jinxin from Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, have engineered a biomimetic composite scaffold (MnLA/HBP) that delivers two gasotransmitters—carbon monoxide (CO) and nitric oxide (NO)—in an inflammation‑responsive manner.
The researchers used cryogenic 3D printing to fabricate the scaffold. Low temperature preserves the activity of heat‑sensitive gas prodrugs: manganese carbonyl (MnCO) as a CO donor and L‑arginine as an NO donor. The resulting scaffold features a highly interconnected porous structure (porosity ~70%, pore size 100–500 μm) and a compressive strength of about 13 MPa, matching human cancellous bone. More importantly, the scaffold releases CO and NO only when triggered by reactive oxygen species (H2O2) in the inflammatory microenvironment—avoiding premature leakage and off‑target toxicity.
In vitro mechanistic studies revealed a well‑orchestrated cascade. CO simultaneously inhibits the NF‑κB pathway and activates the Nrf2 pathway, reprogramming macrophages from a pro‑inflammatory M1 phenotype to a pro‑regenerative M2 phenotype. This shift dramatically reduces inflammatory cytokines (IL‑6, iNOS) and elevates anti‑inflammatory markers (IL‑10, Arg‑1). Meanwhile, NO activates the MAPK/ERK and PI3K‑Akt pathways in endothelial cells, promoting cell migration and tube formation. The two gases also synergistically activate the sGC‑cGMP‑PKG axis in osteoblasts, significantly up‑regulating osteogenic genes (ALP, Runx2, OCN, OPN) and enhancing matrix mineralization.
The researchers then tested the scaffold in a rat critical‑sized calvarial defect model (5 mm diameter). After 12 weeks, micro‑CT analysis showed that the MnLA/HBP scaffold achieved a bone volume/tissue volume (BV/TV) ratio of over 60%, far outperforming single‑drug scaffolds (MnCO/HBP or L‑Arg/HBP) and blank controls. Histological staining (H&E, Masson) confirmed robust new bone formation, dense collagen deposition, and mature mineralized tissue. Immunohistochemistry revealed strong expression of osteocalcin (OCN) and osteopontin (OPN) in the MnLA/HBP group. Importantly, no toxicity was detected in major organs (heart, liver, spleen, lung, kidney), confirming excellent biocompatibility.
This study establishes a new “immune‑vascular‑osteogenic” sequential therapy paradigm through gasotransmitter synergy. The cryogenic 3D printing strategy not only preserves the activity of labile gas prodrugs but also allows precise control over scaffold architecture. The MnLA/HBP scaffold offers a highly promising platform for treating critical‑sized bone defects, with potential for clinical translation.
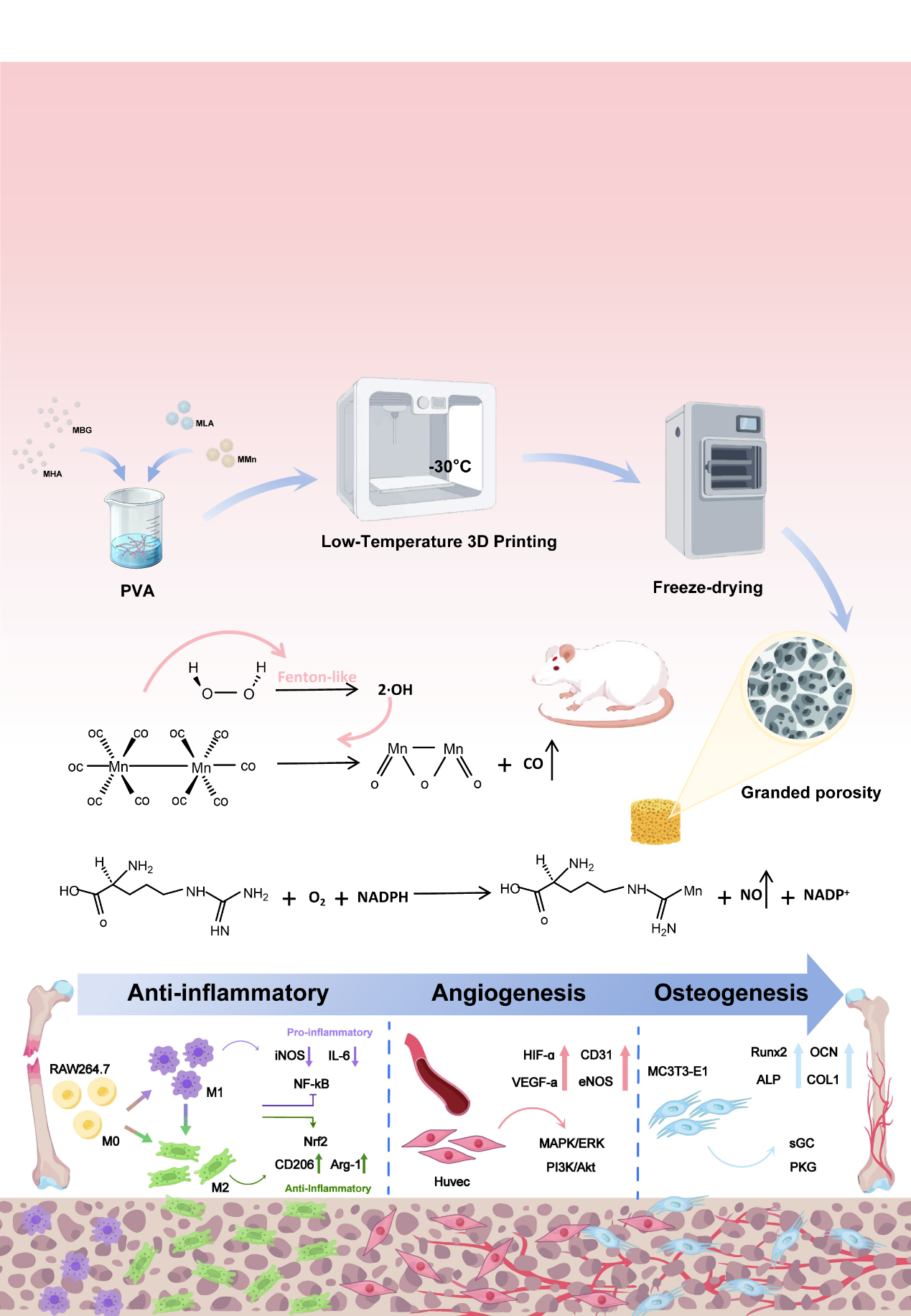
Illustration of the Research(Image by Prof. LIN’s group)
Contact:
Prof. LIN Jinxin
Fujian Institute of Research on the Structure of Matter
Chinese Academy of Sciences
Email: Franklin@fjirsm.ac.cn